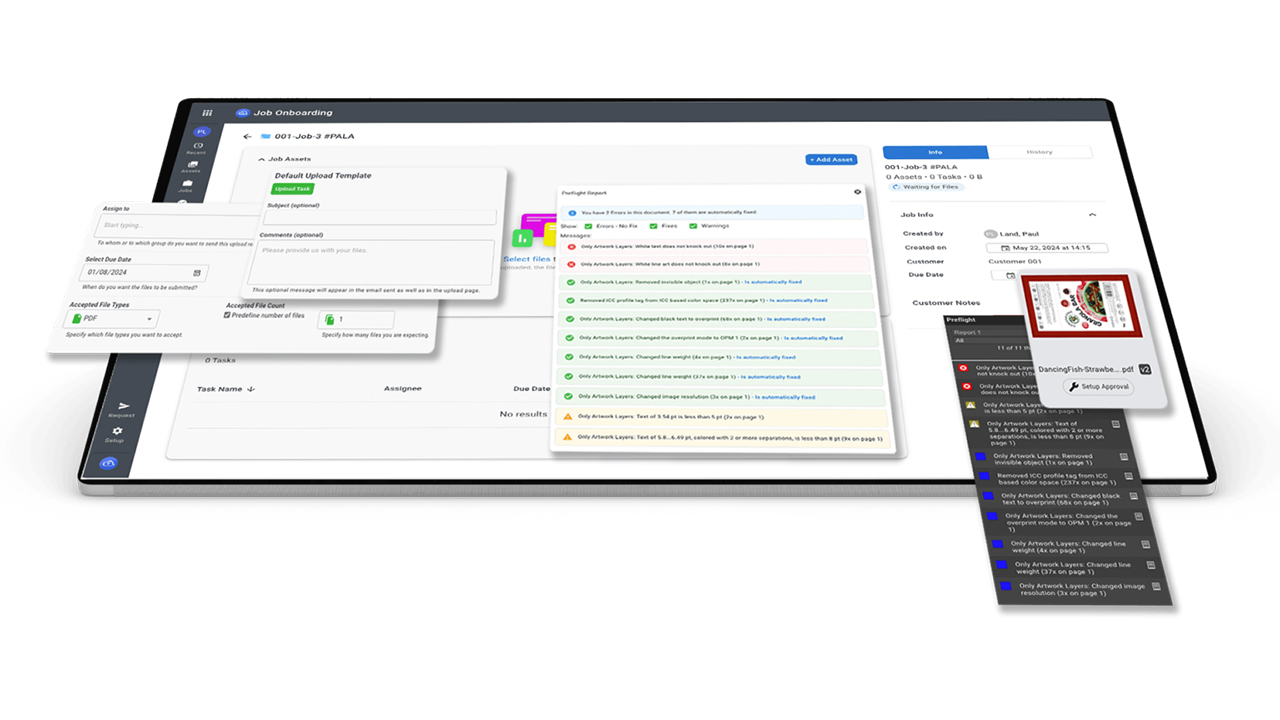
Dion Label registers with FDA, EU Annex 11
Dion Label Printing is now registered with the FDA and compliant with both FDA 21 CFR Part 11 and EU Annex 11.

Part 11 was established in 1997 by the Food and Drug Administration to regulate security of electronic records. The law was established for pharmaceutical, medical and similar FDA-regulated biotech companies. As a label printer for those industries, Dion Label Printing was required to complete the process to be a compliant printer and provide guaranteed secure printing.
Dion Label is a digital and flexographic label printer located in Westfield, Massachusetts.
EU Annex 11 was established in 1992 through the European Union and details the steps needed to maintain Good Manufacturing Practices (GMP) in relation to electronic records and systems. GMP helps maintain control over items produced to confirm quality is maintained. The law ties directly into Part 11 as quality and compliance is especially necessary in pharmaceutical and medical companies.
Co-owner John Dion said: ‘As the FDA ramps up enforcement under 21 CFR Part 11, our compliance (and also with EU Annex 11) will provide select customers greater confidence in computer software, system and records security at Dion Label as required under the rules. Although the process is initially time-consuming, maintenance of our status will have minimal effect on the cost of product manufactured accordingly.’
Stay up to date
Subscribe to the free Label News newsletter and receive the latest content every week. We'll never share your email address.