Schreiner MediPharm launches vial security label
Cap-Lock for Vials combines irreversible tamper evidence with RFID and NFC digital tracking capabilities.
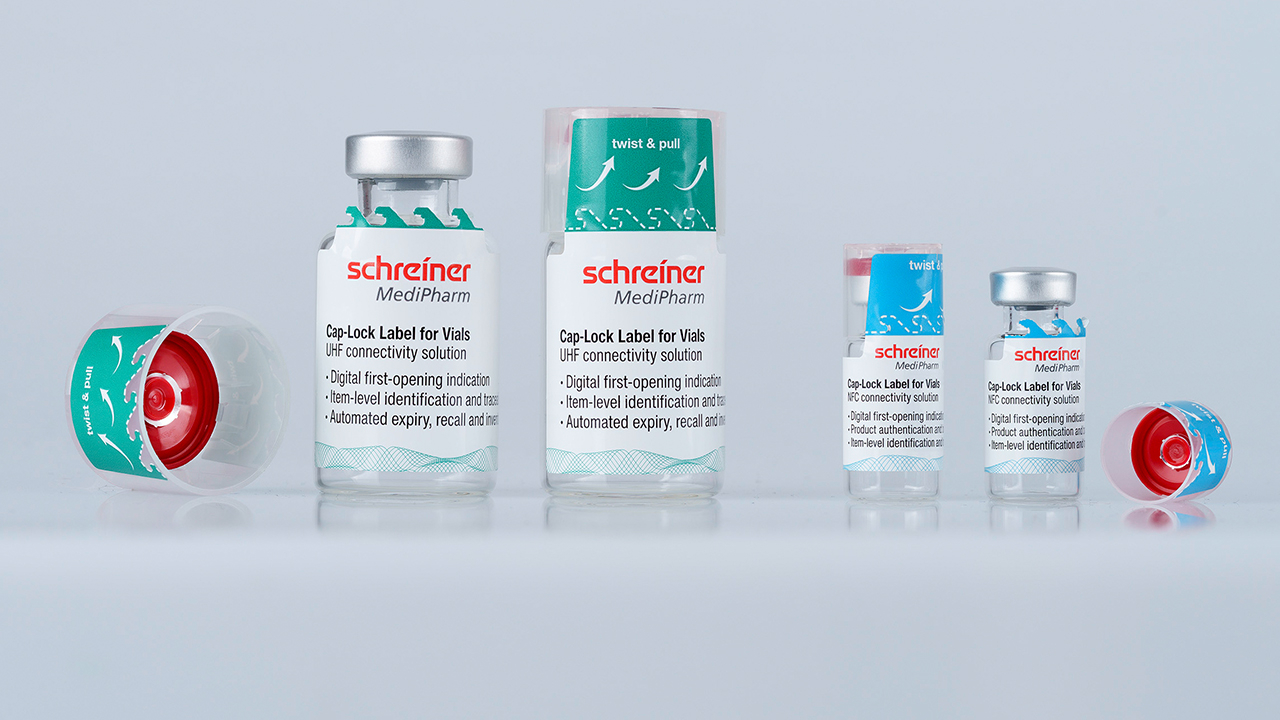
Schreiner MediPharm has launched Cap-Lock for Vials, a functional label developed in cooperation with CDMO OechslerHealth that provides both analog and digital first-opening indication for pharmaceutical vials, addressing product integrity and supply chain transparency challenges.
The system pairs a plastic cap designed by OechslerHealth that permanently interlocks with a vial's flip-off cap with a Schreiner MediPharm label applied to both components. When the vial is opened, the label is irreversibly destroyed, making tampering clearly visible and preventing undetected reapplication. The product is adaptable to all commonly used vial sizes.
Beyond the physical tamper-evidence, Cap-Lock for Vials incorporates an RFID chip that enables a digital first-opening indication and unit-level tracking, allowing previously opened containers to be automatically identified in medication management environments such as hospitals. An NFC function also supports smartphone-based product authentication for brand protection.
Cap-Lock for Vials was presented publicly for the first time at Pharmapack in Paris. Schreiner MediPharm says the product supports pharmaceutical manufacturers in protecting vial integrity from the filling process through to patient administration, combining analog security with digital functionality across the pharma supply chain.
Stay up to date
Subscribe to the free Label News newsletter and receive the latest content every week. We'll never share your email address.

