★You are viewing this content as a subscriber
★Subscribers only
Flexible packaging: inks, coatings, curing and laminating
The role of inks, coatings, curing, varnishing and laminating in the narrow-to-mid-web flexible packaging sector
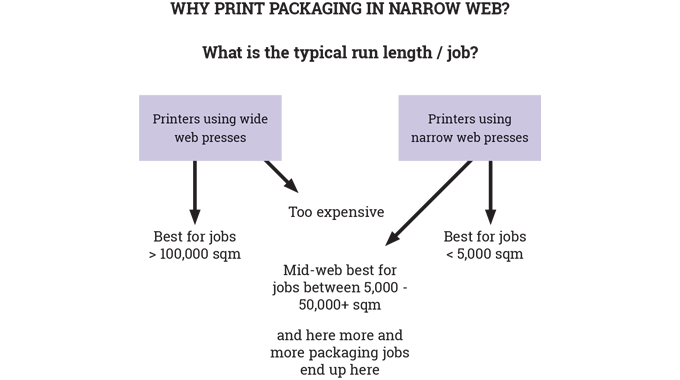
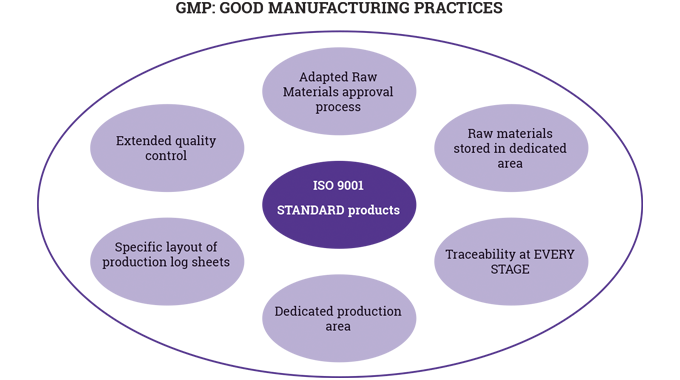
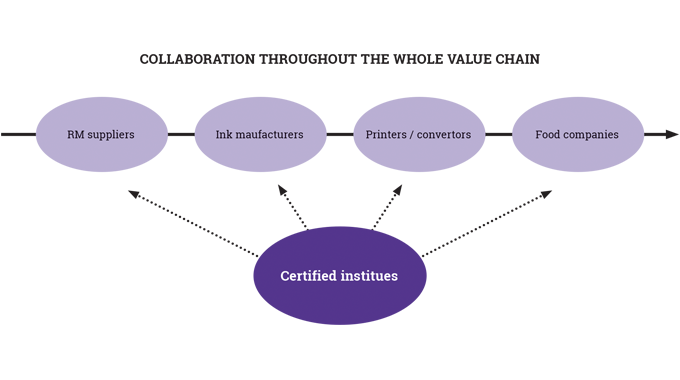
So far in these series of articles, it has been possible to cover a number of key areas in relation to the production of flexible packaging: materials, pre-press, and the printing processes, both conventional and digital. What has not so far been discussed in any detail is the nature and role of inks, coatings, curing, varnishing and laminating used in the narrow-to-mid-web flexible packaging sector.
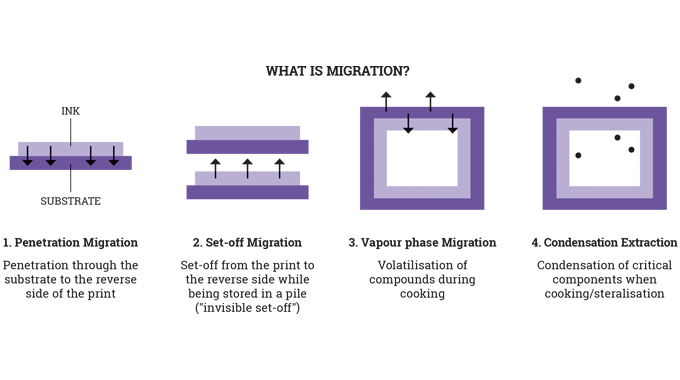
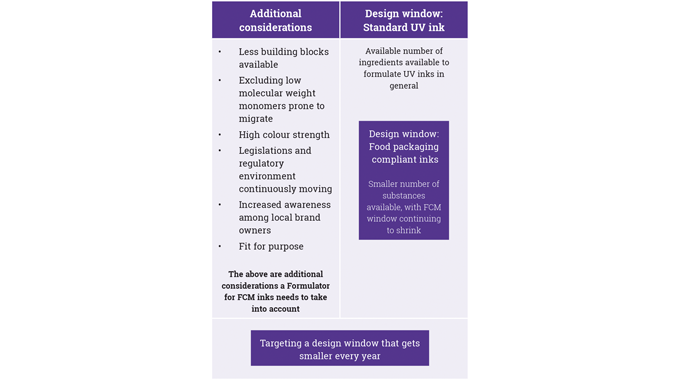
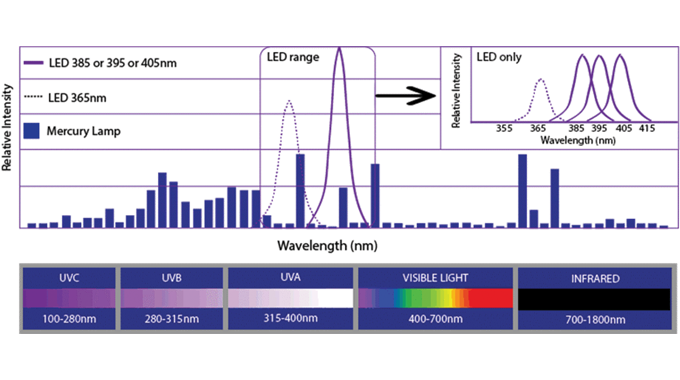
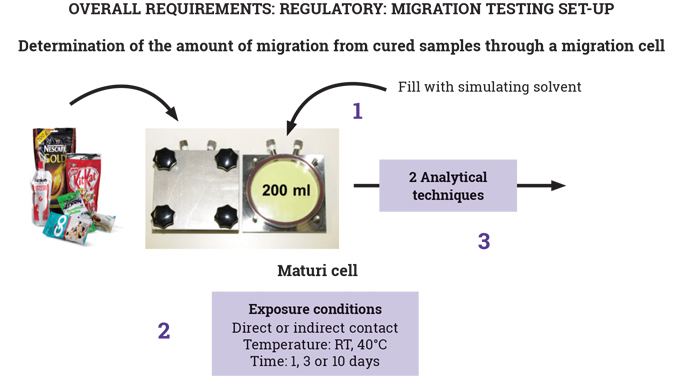
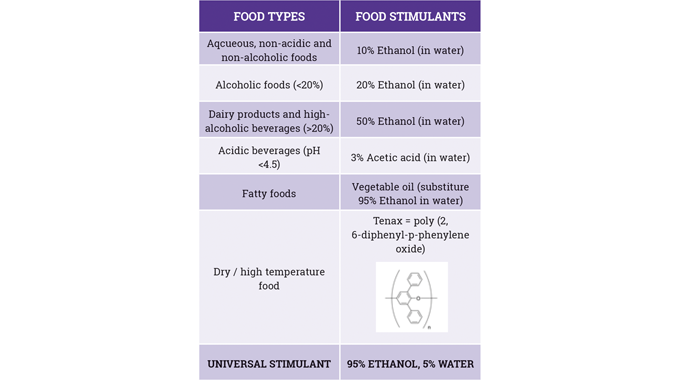
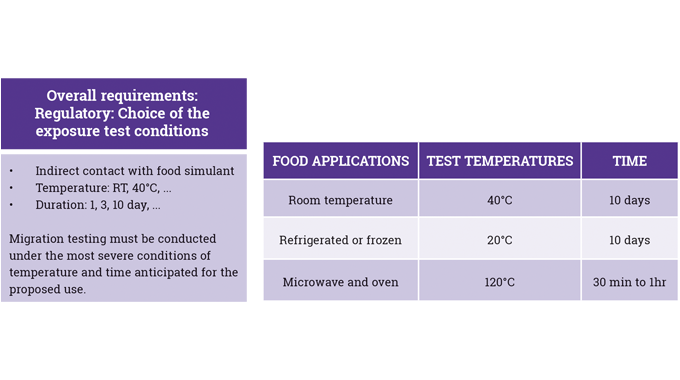
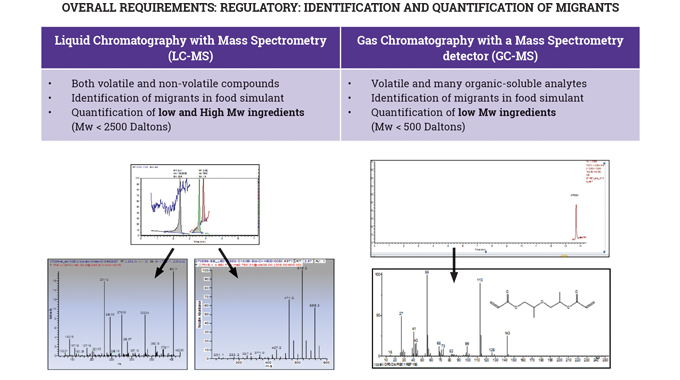
In particular, the use of UV-curable inks and the overall requirements of UV-curable food contact material (FCM) inks in terms of:
Stay up to date
Subscribe to the free Label News newsletter and receive the latest content every week. We'll never share your email address.