Pressure-sensitive adhesive technologies
Adhesive is at the core of the pressure-sensitive label. This article looks at what adhesive is, how it works, why it sometimes fails and how to make sure that it keeps working in often hostile end use environments.
ADHESIVE PROPERTIES
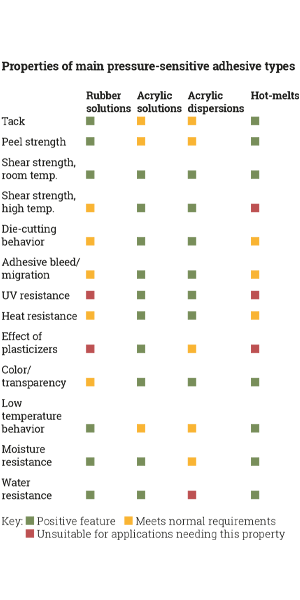
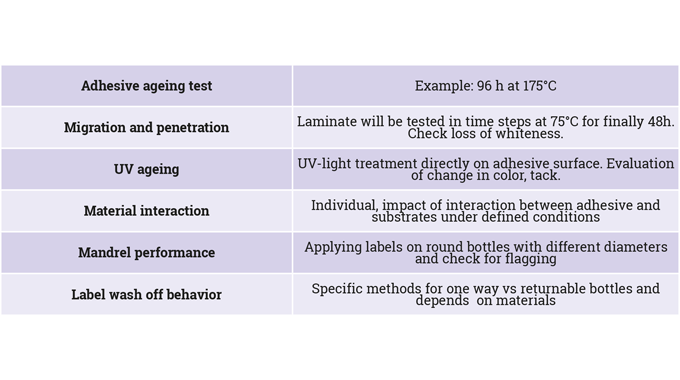
Descriptions of permanent adhesives talk about how well they stick well to certain surfaces – for example ice bucket resistance, BS 5609 sea water resistance, or adhesion at low or very high temperatures. Also important is the type of application – for example a requirement for clarity on a no-look-label construction, or for regulatory compliance on pharmaceutical or indirect food contact applications.
Stay up to date
Subscribe to the free Label News newsletter and receive the latest content every week. We'll never share your email address.