Automating ink logistics
Ink accounts for about six percent of the operational costs of a typical label printing business. But that figure greatly underplays the value that controlled, integrated and automated ink logistics brings, not to mention the potential for lost value.
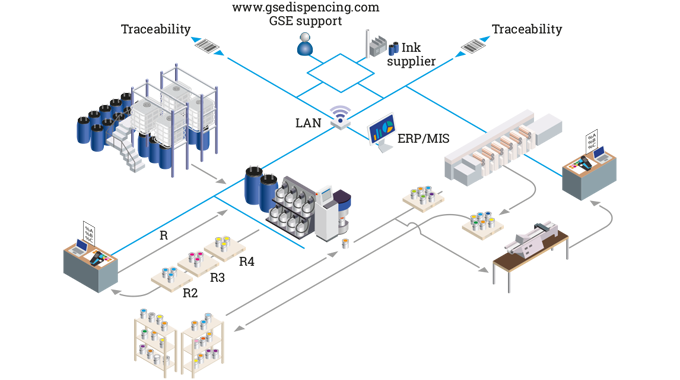
Inks are either supplied pre-mixed to the converting plant, or are mixed on site.
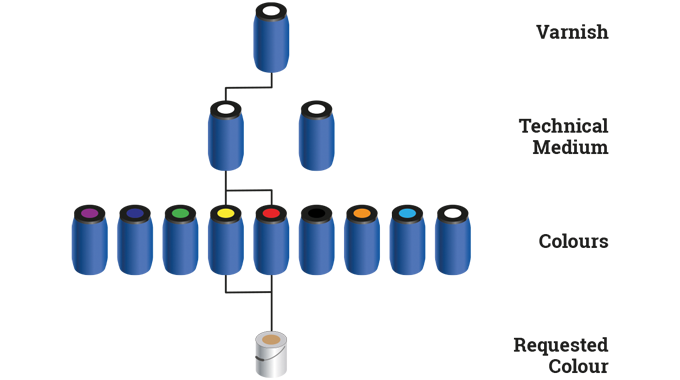
Manual mixing of flexographic inks to match a particular pantone color is a long-established process in the label industry. Inks are weighed out according to ‘recipes’ then the color is tested, either by eye, or with a spectrophotometer and possibly a testing rig, involving a miniature flexo print station (see article on Testing, proofing and quality assurance).
Once at the press, the ink tray is filled manually.
Stay up to date
Subscribe to the free Label News newsletter and receive the latest content every week. We'll never share your email address.