Impact of legislation on ink manufacture and usage
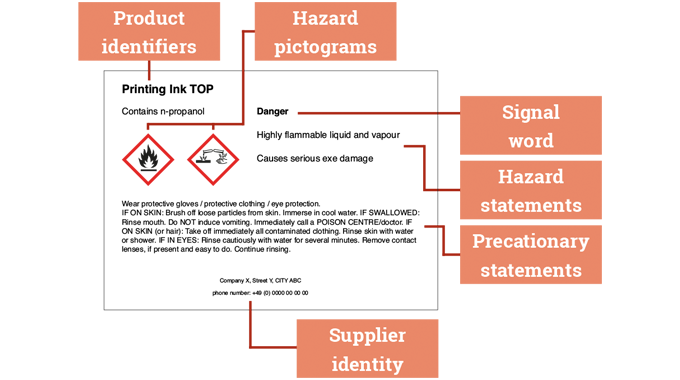
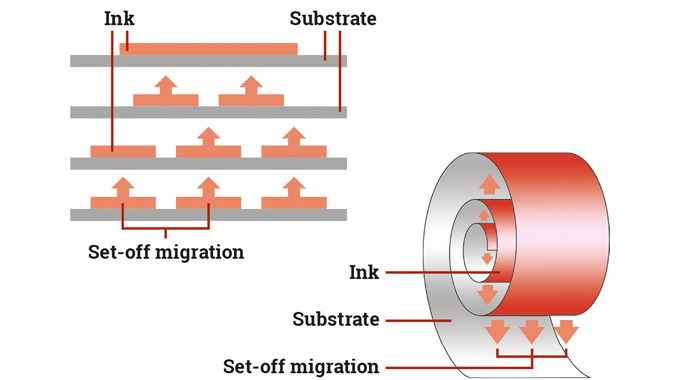
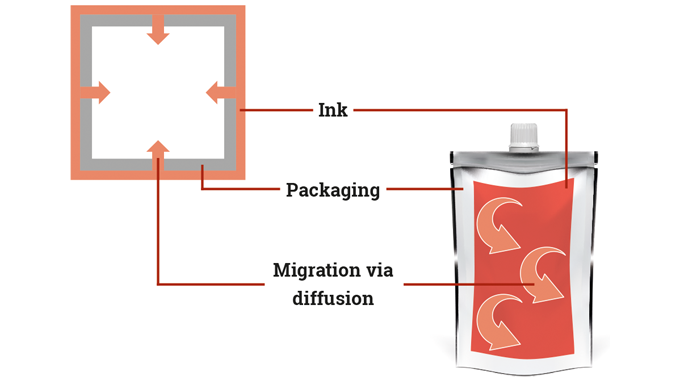
Label and package printing inks, varnishes and coatings are made-up of a complex mix of ingredients. Depending on their particular requirements and construction, they can contain solvents, drying oils, additives, photo-initiators, catalysts and other chemical compounds which, on their own or combined, can contribute or involve potential physical, health or environmental hazards if not handled, stored, or used correctly.
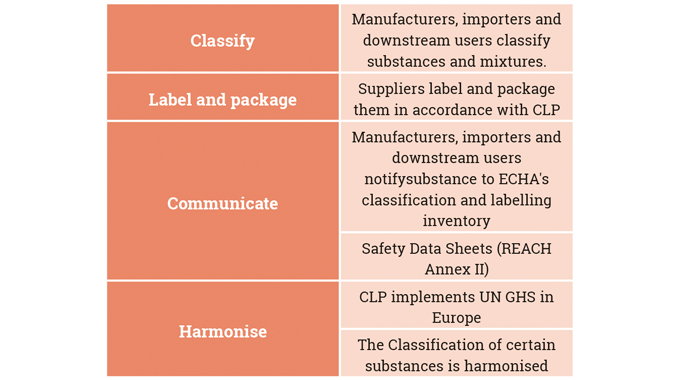
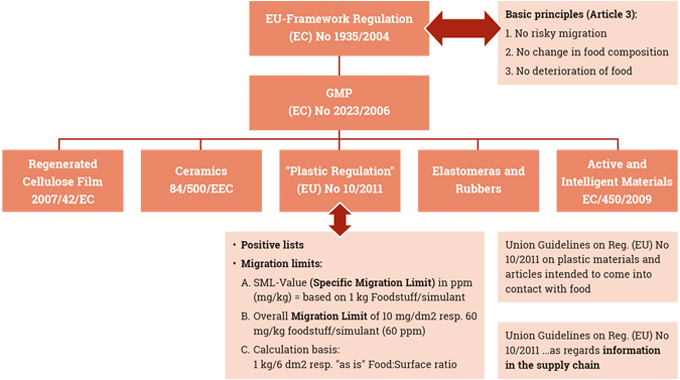
Chemical legislation and food contact legislation are therefore a major influence on the operations of both ink manufacturers and label converters.
Stay up to date
Subscribe to the free Label News newsletter and receive the latest content every week. We'll never share your email address.